
The percentage of the world reserves located in the country with the largest reserves. The higher the value, the larger risk there is to supply. The percentage of an element produced in the top producing country. 
Low = substitution is possible with little or no economic and/or performance impact Medium = substitution is possible but there may be an economic and/or performance impact High = substitution not possible or very difficult. The availability of suitable substitutes for a given commodity. A higher recycling rate may reduce risk to supply. The percentage of a commodity which is recycled. The number of atoms of the element per 1 million atoms of the Earth’s crust. This is calculated by combining the scores for crustal abundance, reserve distribution, production concentration, substitutability, recycling rate and political stability scores. The Chemical Abstracts Service registry number is a unique identifier of a particular chemical, designed to prevent confusion arising from different languages and naming systems.ĭata for this section been provided by the British Geological Survey.Īn integrated supply risk index from 1 (very low risk) to 10 (very high risk). Where more than one isotope exists, the value given is the abundance weighted average.Ītoms of the same element with different numbers of neutrons. This is approximately the sum of the number of protons and neutrons in the nucleus. The mass of an atom relative to that of carbon-12. 
The transition of a substance directly from the solid to the gas phase without passing through a liquid phase.ĭensity is the mass of a substance that would fill 1 cm 3 at room temperature. The temperature at which the liquid–gas phase change occurs. The temperature at which the solid–liquid phase change occurs. The arrangements of electrons above the last (closed shell) noble gas. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. Although there are exceptions, such as former Molecule of the Week krypton difluoride.A vertical column in the periodic table. 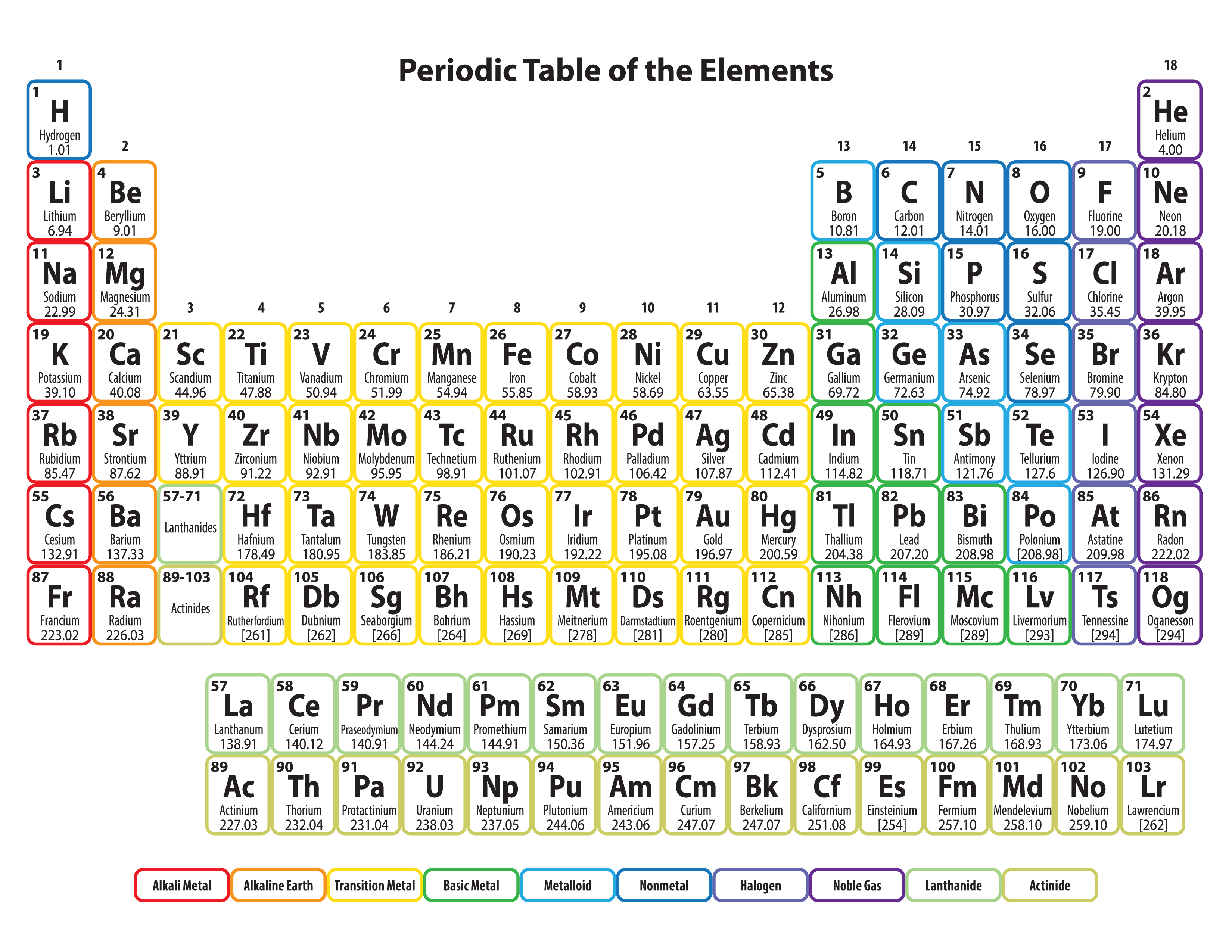
Over the years, the mythological kryptonite was endowed with much more extensive chemistry than real krypton gas.ġ. In comic books dating back to 1949, “Krypton” was the planet where Superman was born and “kryptonite” was the only substance that the superhero was vulnerable to. At one time, the wavelength of light emitted by the 86Kr isotope was used to define the meter, but in 1983, the meter’s definition was changed to a function of the speed of light. Krypton has few practical applications, mostly in bright white light bulbs used in photography and in devices used in physical and chemical research. Like all inert gases, the boiling and melting points of krypton are only a few degrees apart (see “fast facts”). For his work in the discovery of several inert gases, Ramsay was awarded the Nobel Prize in Chemistry in 1904. In 1898, British chemists William Ramsay and Morris Travers discovered krypton as the residue of evaporating almost all of the other components of liquid air. Its concentration in Earth’s atmosphere is ≈1 ppm by volume. 1 It is the fourth atom in group 18 of the periodic table of elements, after helium, neon, and argon. Krypton is an inert (or “noble”) gas and, as such, is not bound up in molecules but exists as single atoms. I’m a real element that has a fictional counterpart.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
